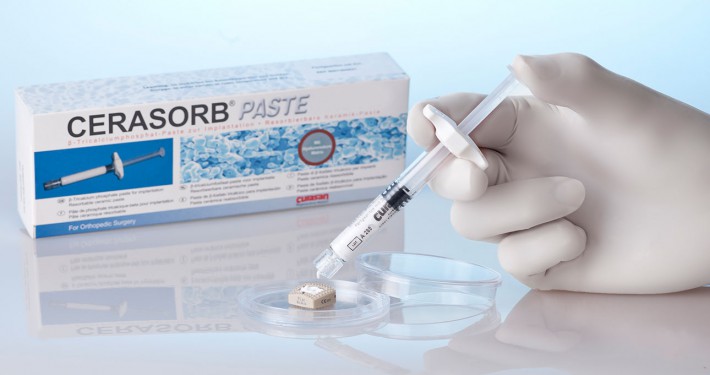
CERASORB
The safest way to new bone
Bone replacement materials are routinely used today in dental medicine for the treatment of bone defects caused by trauma or disease.
The CERASORB® product family provides bone replacement material for this purpose, and this is completely resorbed in the context of bone regeneration. In virtue of its chemical and material properties it provides 100% assurance in defect restoration.
When using the CERASORB® products a risky second procedure for harvesting cancellous bone can usually be avoided. This has the advantage for clinicians and patients that the problem of rejection reactions or potential infection and immunological risks associated with materials of biological origin are eliminated, patient information need not address these risks.
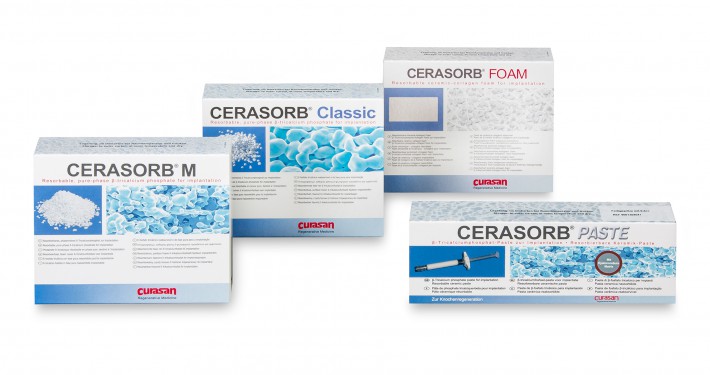
CERASORB® Classic is a resorbable, pure–phase β–tricalcium phosphate–ceramic matrix with open, interconnecting porosity.
CERASORB® M is a further development based on CERASORB® Classic. It has an interconnecting, open multi-porosity with micro-, meso- and macropores (5 μm – 500 μm) and an overall porosity of approximately 65%. The granules are polygonal, i.e., irregularly shaped and enhance intercalation and interlocking in the defect cavity.
The different forms in the CERASORB® product family provide the ideal basis for the treatment of a diverse array of indications.
 En
En Ar
Ar